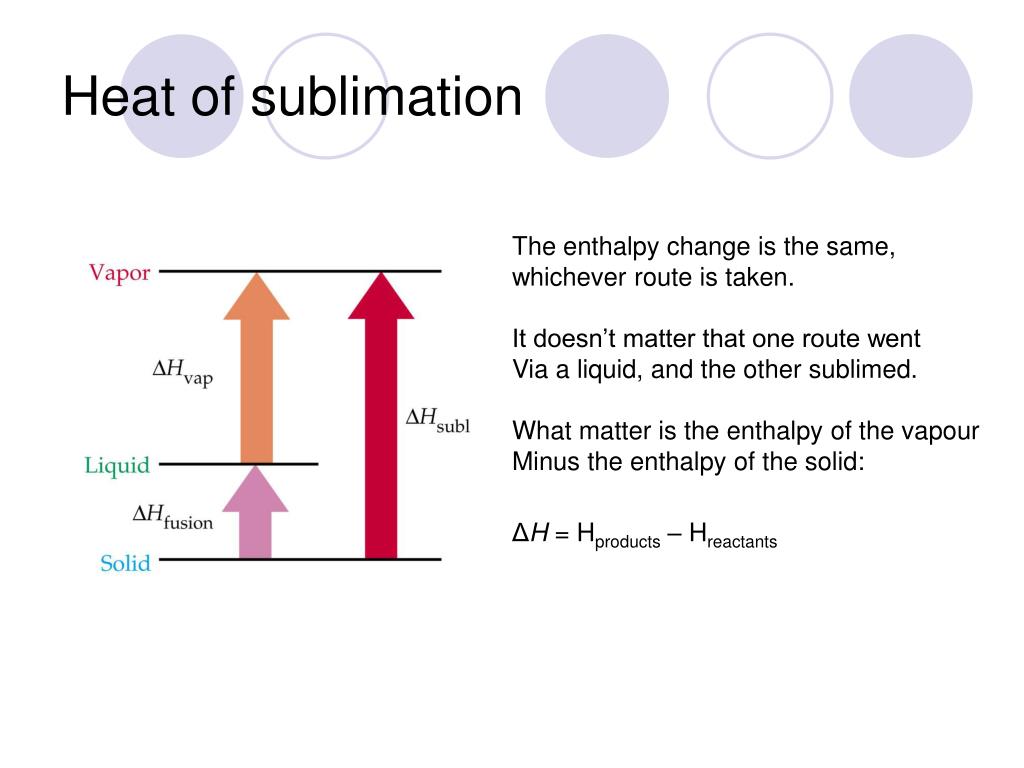
Here is how you would classify the phase changes as endothermic or exothermic:
Is converting frost to water vapor endothermic or exothermic. Melting, evaporation and sublimation are endothermic processes while freezing, condensation. This typically occurs when water vapor molecules come. An exothermic reaction gives off heat energy.
Therefore, boiling water is an endothermic process. Condensation is the process by which water vapor turns into liquid water. Thus dissolution of a gas in water is an exothermic process.
The molar heat of condensation is the heat evolved when one mole of vapor is condensed. The applied heat helps break the intermolecular bonds between the water molecules, which spreads in the molecules further. An endothermic process absorbs heat and cools the surroundings.
Heat is evolved, partly in consequence of the chemical combination and partly from the condensation of the gas to the. In an exothermic reaction, energy is released in the form of heat.









