Hclo, hclo2, hclo3, hclo4 according to bronsted lowery concept, a strong acid has weak.
Which of the following is the strongest acid. Which of the following is the strongest acid? This compound is known to be the second strongest acid of hydrogen and a halogen (the strongest being hydrostatic acid). (a) clch 2 cooh (b).
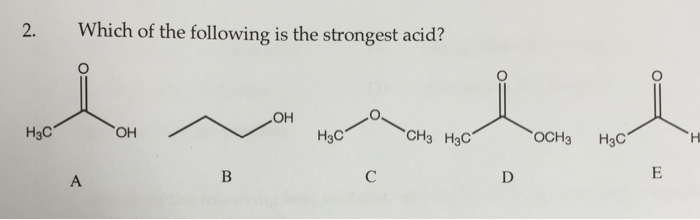
Testbank, question 028a which one of the following is the strongest acid? Which of the following is the strongest acid? Amongst the following compounds the strongest acid is :
List of strong acids there are 7 strong acids: Which of the following is strongest acid ? Which of the following is a strongest acid among oxy acids of chlorine and why?
So it is more acidic. Which of the following is the strongest acid? Correct option is c) (c) hclo 4 reason :
Among the following compounds the strongest. Cooh so,h (a) o oh ch,oh. Which of the following is strongest acid?
Bromoacetic acid fluoroacetic acid chloroacetic acid acetic acid this problem has been solved! A b c d medium solution verified by toppr correct option is c a strong acid has a weak conjugate base. All the acids have the same strength.









