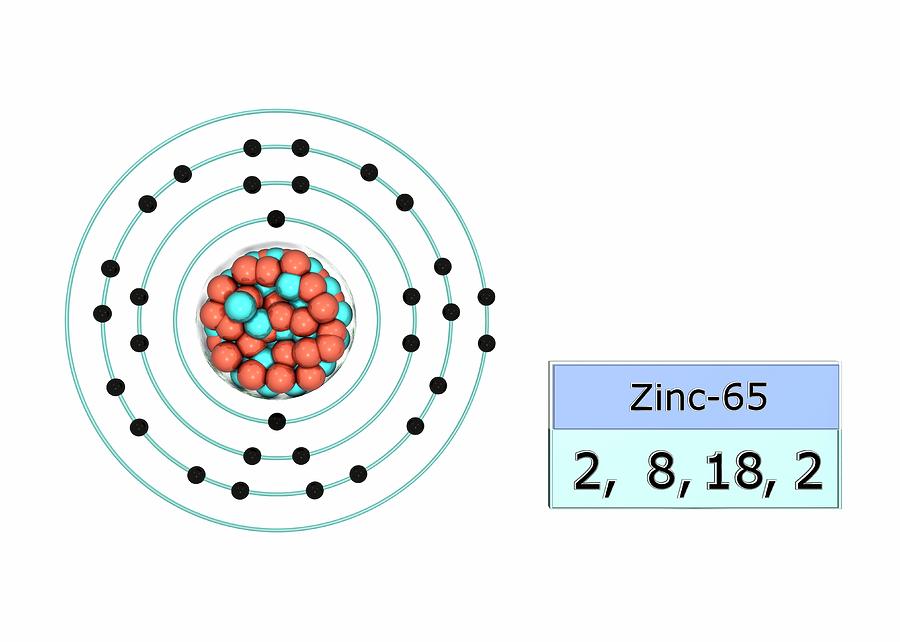
The electron configuration for zinc is 1s2 2s2 2p6 3s2 3p6 4s2 3d10.
What is the electron configuration for zinc. Ground state electron configuration of zinc (zn):. Zn (zinc) is an element with position number 30 in the periodic table. The electron configuration of a neutral zinc atom is 1s 22s 22p 63s 23p 63d.
The chemical symbol of zinc is 'zn'. Zinc 30 is a very stable metal. In general, the electronic configuration of these elements.
6 rows zinc has an electron configuration of [ar]3d 10 4s 2 and is a member of the group 12 of the. 5 rows the zinc atom donates two electrons in the 4s orbital to form a zinc ion (zn 2+ ). Chemistry electron configuration electron configuration.
The electronic configuration of zinc is 1s2,2s2,2p6,3s2,3p6,4s2,3d10. What is zinc's electron configuration? Electron configuration chart of all elements is mentioned in the table below.the shorthand electron configuration (or noble gas configuration) as well as full.
1 answer anor277 may 24, 2016. It is stable in the presence of oxygen, but it is unstable in low oxygen conditions. Represented in the periodic table as zn, zinc is a transition metal, grouped with cadmium and.
Zinc is a chemical detail with the symbol zn and atomic number 30. The 2+ charge denotes that zinc has loosened 2 electrons from its valence shell. What is the ground state electron configuration for zinc?








