Specific heat capacity is the amount of heat energy required to raise the temperature of a substance per unit of mass.the specific heat.
What is heat capacity. Specific heat capacity refers to a. C) is the amount of heat in joules. We now introduce two concepts useful in describing heat flow and temperature change.
Specific heat capacity definition [ edit]. Heat capacity is an intrinsic physical property that determines the level of heat required to change a substance’s temperature by a predetermined amount. It can be defined as the amount of heat to be supplied to an object in order to produce a unit change.
We can define heat capacity as the amount of heat required to raise the temperature of a given mass of substance by 1 kelvin (or 1 ℃). Specific heat is defined by the amount of heat needed to raise the temperature of 1 gram of a substance 1 degree celsius (°c). The corresponding intensive property is the specific heat capacity, found by dividing the heat capacity of an object by.
Heat capacity (usually denoted by the capital c, often with subscripts), or thermal capacity, is the measurable physical quantity that characterizes the amount of heat required to change a. Heat capacity has the units of energy per degree. Heat capacity is also known as thermal capacity and it is defined as the amount of heat required to raise the temperature of a given mass of the substance by one.
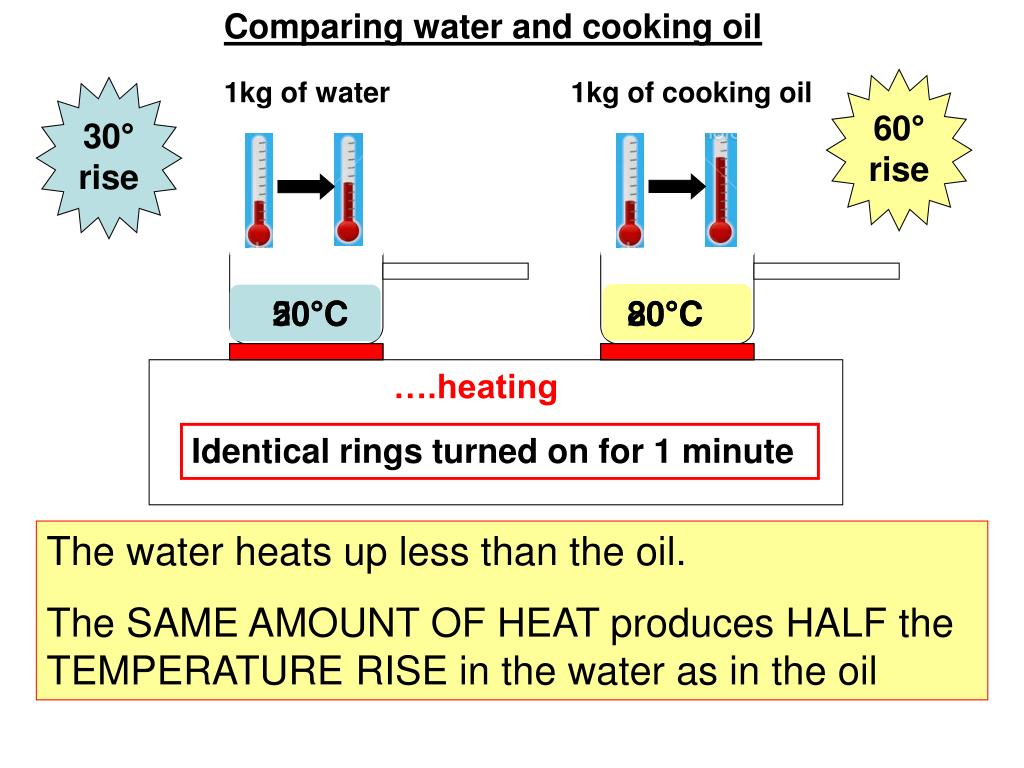
Heat capacity the specific heat capacity of water is 4,200 joules per kilogram per degree. Heat capacity is calculated by the formula: It is denoted by c and is an.
Where, q is the amount of heat required in joules. Heat capacity is an extensive property. Q = cδ δ t.









/chemistry-experiment-172594210-571b7b5a3df78c5640f730fa.jpg)