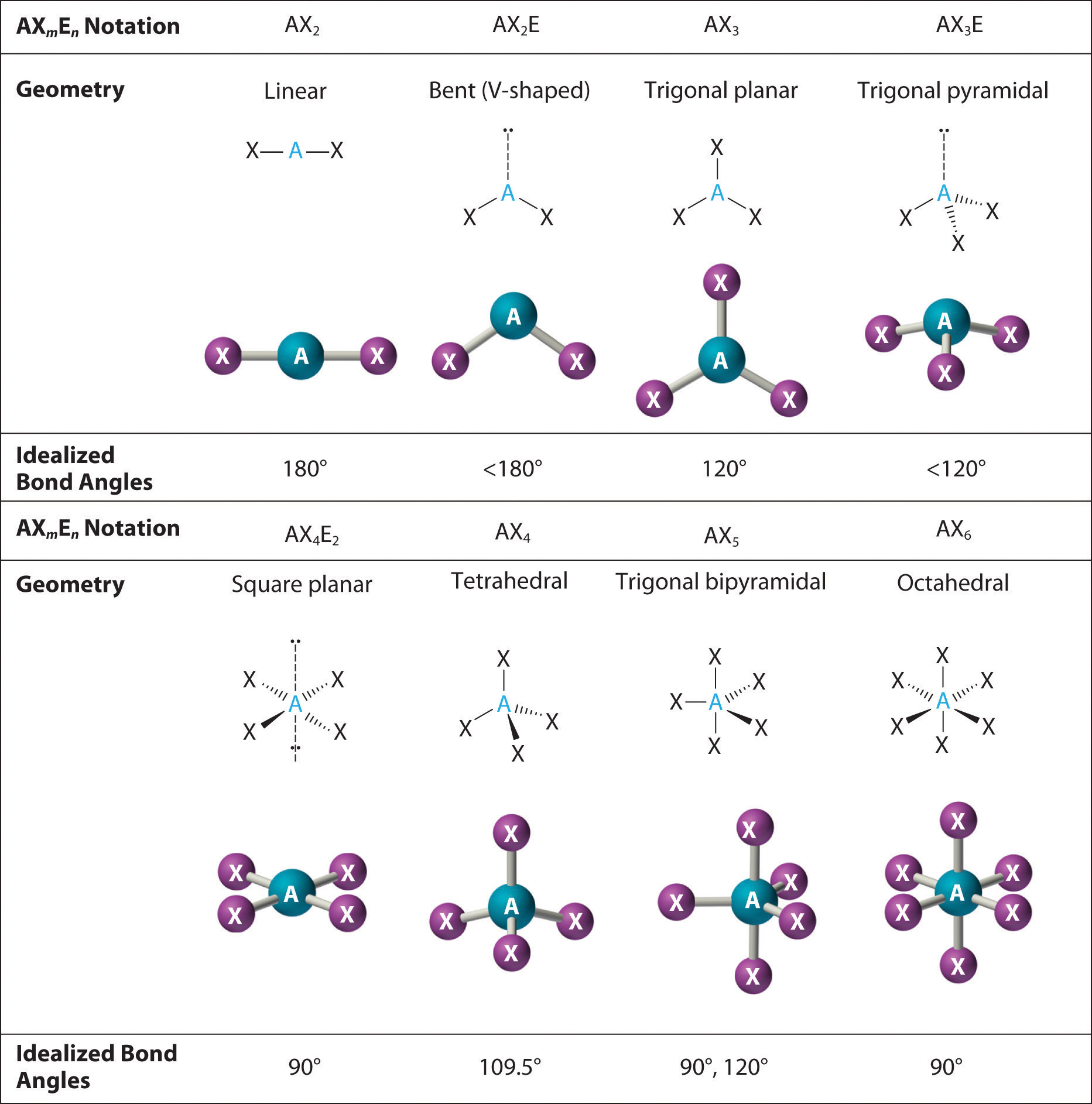
3 electron pairs, trigonal planar;
How to draw 3d vsepr models. 3d vsepr app helps you to visualize the shapes of the vsepr models in 3d such that you can understand more and you can sort out your confusions. If it helps when drawing the 3d aspects, think of the rectangle as a long, stretched out cube. How does molecule shape change with different numbers of bonds and electron pairs?
Web the vsepr model stands for valence shell electron pair repulsion model. Then, draw another square above it and to the right. Web start by drawing a rectangle as the tabletop.
Web the valence shell electron pair repulsion theory (vsepr) helps us to understand the 3d structure of molecules. It doesn’t matter which side you chose. Find out by adding single, double or triple bonds and lone pairs to the central atom.
Next, connect the corners of the first square to the corners of the second. Web representing three dimensions on 2d paper (or computer screen) is challenging. The vsepr model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom.
Web i can identify and draw the 3d shape of molecules using vsepr theory. Types of vsepr structures include linear, trigonal planar and tetrahedral. Web the valence shell electron pair repulsion (vsepr) theory is a simple and useful way to predict and rationalize the shapes of molecules.
Then, draw four cylinders or 3d poles coming from each of the four sides. This education app help the students to learn chemistry in a smarter way. Draw 3d shapes by applying the basic rules of linear perspective.



















