The shorthand noble gas electronic configuration of zn is [ar].
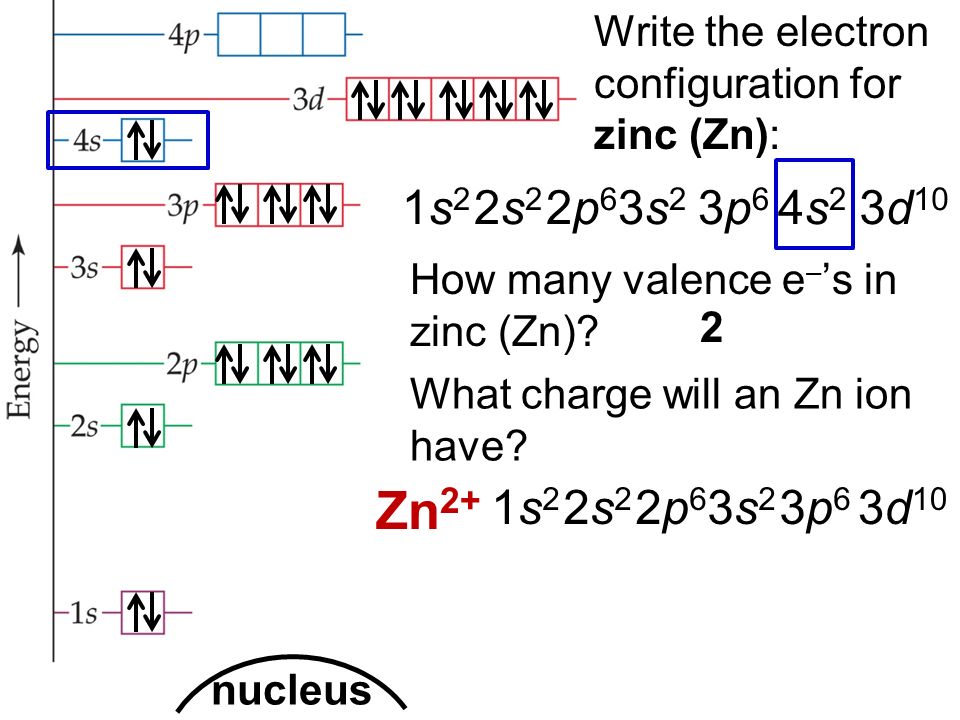
Ground state electron configuration for zn. The zinc atom donates two electrons in the 4s orbital to form a zinc ion (zn 2+ ). The correct electron configuration of zn 2+ is 1s2 2s2 2p6 3s2 3p6 3d10 or. The symbol of the element is zn.
The atom is electrically neutral. Total = 4 + 4 + 8 = 1 6 electrons. Ge, fe, zn, ni, w, ti.
Write the ground state electron. Therefore, the number of protons is equal to the number of electrons. It states that it has 30.
The ground state is where electrons are in the lowest possible energy while the excited state is where the electron is excited and jumps into a higher orbital. We first need to find the nu. Here, the electron configuration of zinc ion (zn 2+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10.
What is the ground state electron configuration for zn? Thus, the ground state electron configuration of gaseous neutral zinc in its ground state is [ar]. 100% (4 ratings) transcribed image text:
To write the configuration for the zinc and the zinc ion, first we need to write the electron configuration for just zinc (zn). (a)[ar]4s13d 10 (b) [ar]3db (c) [ar]4s23d (d) [kr]4s23f1 (e) none of these 16. Which of the following elements is chemically.









