Copper ← zinc → gallium.
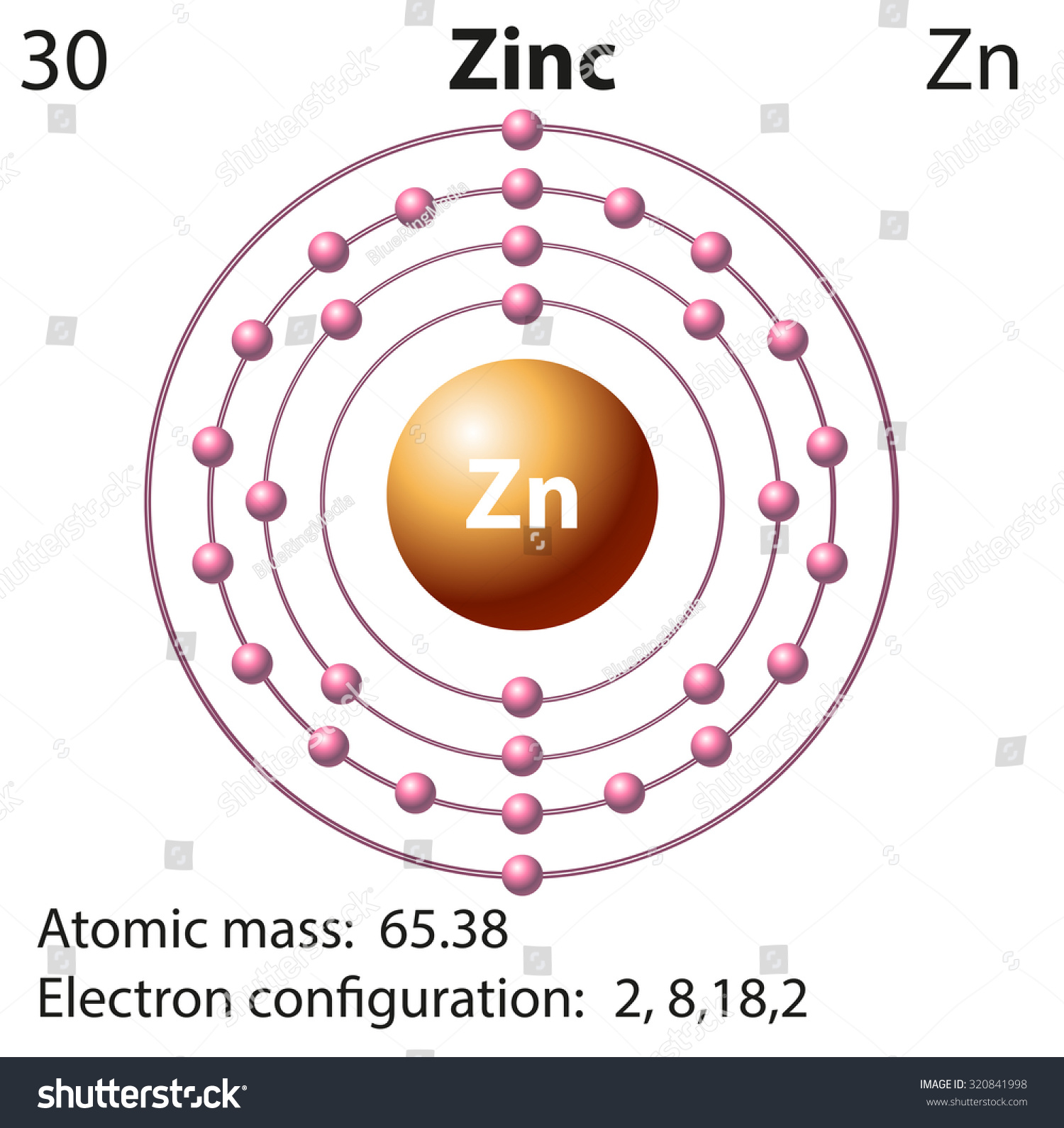
Electron configuration of zinc ion. The electronic configuration of each element is decided by the aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. Zinc's complete electron configuration is 1s22s22p63s23p64s23d10 what is selenium ion electron configuration? 1s2 2s2 2p6 3s2 3p6 3d10 4s2.
1s2 2s2 2p6 3s2 3p6 3d10 4s2. Selenium ion electron configuration is. 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s < 5f < 6d < 7p.
Full electron configuration of zinc: Electronic configuration of zinc ion zn 2 +: The electron configuration of a neutral zinc atom is 1s22s22p63s23p63d104s2.
The electronic configuration of zinc is 1s2,2s2,2p6,3s2,3p6,4s2,3d10. Zn loses its two electrons and results in the formation of zn 2 + ion. The zinc atom donates two electrons in the 4s orbital to form a zinc ion (zn 2+ ).
All electrons are paired, and therefore zn (0) is diamagnetic. Represented in the periodic table as zn, zinc is a transition metal, grouped with cadmium and. The zn2+ ion has lost two electrons, which leaves it with 30 protons and 28 electrons.
The zinc atom donates two electrons in the 4s orbital to form a zinc ion(zn 2+). Zn (zinc) is an element with position number 30 in the periodic table. The same rule will apply to transition metals when forming ions.









